Dr. Leili Rohani is a Scientist II at the MIT Synthetic Biology Center in the Department of Biological Engineering
We develop computationally guided synthetic biology approaches to program cellular decision-making for disease modeling and therapy. Our goal is to engineer cells that can sense pathological states and execute designed responses, with a focus on reprogramming cell states toward healthier phenotypes using in silico modeling and targeted perturbation strategies.

We develop computationally guided synthetic biology approaches to program cellular decision-making for disease modeling and therapy. Our goal is to engineer cells that can sense pathological states and execute designed responses, with a focus on reprogramming cell states toward healthier phenotypes using in silico modeling and targeted perturbation strategies.

We engineer “SMART” organoids that act as context-aware biosensors, dynamically sensing their microenvironment and producing measurable responses that improve the accuracy and predictive power of drug testing.
We develop patient iPSC–derived engineered heart tissue (EHT) platforms to model cardiac arrhythmias, recapitulate disease-relevant electrophysiological phenotypes, and create instrumented tissue systems to study how inflammation and other stressors influence cardiac dysfunction.
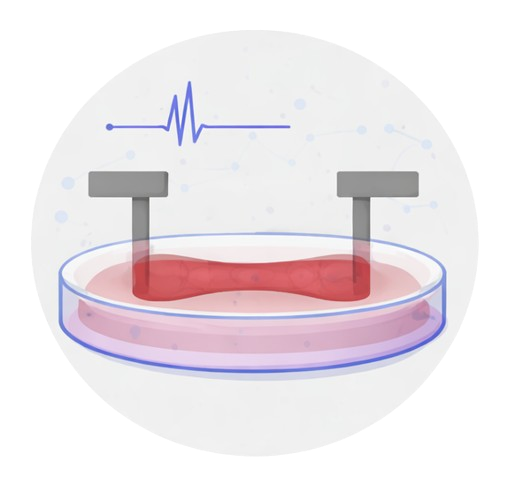
We develop patient iPSC–derived engineered heart tissue (EHT) platforms to model cardiac arrhythmias, recapitulate disease-relevant electrophysiological phenotypes, and create instrumented tissue systems to study how inflammation and other stressors influence cardiac dysfunction.

We use bioprocess engineering and cell manufacturing strategies to scale the production of clinically relevant human pluripotent stem cells (hPSCs) in bioreactors, supporting applications in cell therapy and regenerative medicine.
We develop patient iPSC–derived engineered heart tissue (EHT) platforms to model cardiac arrhythmias, recapitulate disease-relevant electrophysiological phenotypes, and create instrumented tissue systems to study how inflammation and other stressors influence cardiac dysfunction.

We develop patient iPSC–derived engineered heart tissue (EHT) platforms to model cardiac arrhythmias, recapitulate disease-relevant electrophysiological phenotypes, and create instrumented tissue systems to study how inflammation and other stressors influence cardiac dysfunction.

We use bioprocess engineering and cell manufacturing strategies to scale the production of clinically relevant human pluripotent stem cells (hPSCs) in bioreactors, supporting applications in cell therapy and regenerative medicine.
We develop patient iPSC–derived engineered heart tissue (EHT) platforms to model cardiac arrhythmias, recapitulate disease-relevant electrophysiological phenotypes, and create instrumented tissue systems to study how inflammation and other stressors influence cardiac dysfunction.

We develop patient iPSC–derived engineered heart tissue (EHT) platforms to model cardiac arrhythmias, recapitulate disease-relevant electrophysiological phenotypes, and create instrumented tissue systems to study how inflammation and other stressors influence cardiac dysfunction.

We use bioprocess engineering and cell manufacturing strategies to scale the production of clinically relevant human pluripotent stem cells (hPSCs) in bioreactors, supporting applications in cell therapy and regenerative medicine.
Schedule a consultation to discuss your genetic health concerns and explore personalized solutions.